Your Project. Our Priority.
Hart Clinical Consultants (HCC) was founded in March 2011 to support medical device companies with experienced staff for clinical trial execution. As industry demands evolved, so did HCC. Today, HCC operates as a full service Clinical Research Organization (CRO), delivering end to end solutions across every phase of clinical trial operations.
What truly sets HCC apart is a steadfast commitment to three core principles:
1. Excellence in execution
2. Specialized therapeutic expertise
3. Highly personalized client experience
These values ensure that every partner receives the focused attention and dedicated service embodied in our promise:
Your Project. Our Priority.
Leadership Team
The people who drive our mission forward

Jim Hart

Jim Hart, founder, serves as the President and CEO of Hart Clinical Consultants (HCC), LLC. He brings an extensive background in medical device sales and clinical research, supported by more than 30 years of industry experience. Jim’s career spans leadership of a high volume cardiac and endovascular interventional lab, as well as key roles within multiple sponsor organizations prior to establishing HCC. He achieved notable success at legacy companies including Advanced Cardiovascular Systems (ACS), Guidant Corporation, and Abbott Vascular. Jim’s leadership is grounded in strong strategic planning and an unwavering commitment to excellence. His dedication to advancing medical device innovation continues to fuel the growth and success of HCC. He holds a BS degree from the University of Central Florida and is a Certified Clinical Research Associate, recognized by the Association of Clinical Research Professionals.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Suspendisse varius enim in eros elementum tristique. Duis cursus, mi quis viverra ornare, eros dolor interdum nulla, ut commodo diam libero vitae erat. Aenean faucibus nibh et justo cursus id rutrum lorem imperdiet. Nunc ut sem vitae risus tristique posuere.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Suspendisse varius enim in eros elementum tristique. Duis cursus, mi quis viverra ornare, eros dolor interdum nulla, ut commodo diam libero vitae erat. Aenean faucibus nibh et justo cursus id rutrum lorem imperdiet. Nunc ut sem vitae risus tristique posuere.

Jason Krzeszak

Jason serves as Executive Vice President at HCC, leading the Clinical Research and Operations teams with a strategic, evidence-driven approach. With 20 years of experience in the medical device industry, he brings deep expertise across clinical research, regulatory strategy, and preclinical development. Jason has held leadership roles in both startups and multi-product organizations such as Medtronic, consistently delivering clinical programs that support global regulatory approvals and market access. Known for his analytical rigor and collaborative leadership, Jason partners closely with clients to define strategic objectives and execute clinical initiatives that generate high-quality evidence. His expertise spans clinical trial design, operations, regulatory submissions, cross-functional team leadership, and full-spectrum product development. He has supported a wide range of therapeutic areas, including cardiovascular, endovascular, spine, urology, organ preservation, wound care, and respiratory therapy. Jason holds an MBA from Cornell University and MSE/BSE degrees from the University of Michigan in biomedical and materials science engineering.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Suspendisse varius enim in eros elementum tristique. Duis cursus, mi quis viverra ornare, eros dolor interdum nulla, ut commodo diam libero vitae erat. Aenean faucibus nibh et justo cursus id rutrum lorem imperdiet. Nunc ut sem vitae risus tristique posuere.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Suspendisse varius enim in eros elementum tristique. Duis cursus, mi quis viverra ornare, eros dolor interdum nulla, ut commodo diam libero vitae erat. Aenean faucibus nibh et justo cursus id rutrum lorem imperdiet. Nunc ut sem vitae risus tristique posuere.

Sapna Ghuman

Sapna Ghuman is the Director of Biometrics at HCC, where she provides strategic leadership across data management, database programming, and biostatistics. With over twenty years of experience in the life sciences industry, she brings deep expertise in leading and scaling biometrics functions within pharmaceutical, medical device, and clinical data solutions organizations. Sapna is an expert in Electronic Data Capture (EDC) systems and has overseen database design and data management for complex drug and device studies throughout the clinical development lifecycle. Her experience covers a wide range of study types and therapeutic areas, including cardiology, gastroenterology, oncology, rheumatology, neurology, psychiatry, and pulmonary medicine. Sapna works closely with sponsors to guide decisions on EDC platform selection, biometrics resourcing, and data strategy. She ensures high-quality, compliant biometrics solutions from study startup to database lock. Her leadership of global teams supports HCC in delivering reliable biometrics services. Sapna is a certified clinical data manager and holds an MPH degree in epidemiology from Boston University and a BA degree in psychology from Georgetown University.

Melanie Miller


Mark Hart

Mark Hart serves as Comptroller at HCC, leading the Finance team. With 20 + years of experience prior to HCC in the Retail and Banking industry, Mark manages cash flow including invoicing, payroll, accounts payable, accounts receivables, commercial insurance, as well as some onboarding of new staff. Mark partners with internal and external resources of HCC to make sure financial resources are used efficiently and timely to maximize return on investments. Mark received his B.S.B.A. from the University of Central Florida.

Deb Rodgers

Deb serves as the Manager of Clinical Operations at HCC, where she leads the Operations team composed of Clinical Research Associates (CRAs), Project Coordinators (PCs), and Field Clinical Specialists. She brings more than 25 years of clinical research experience spanning site-level research, pharmaceutical, and the medical device industry. Throughout her career, Deb has held roles as a Research Coordinator, Clinical Research Associate, and Research Operations Manager within an academic medical setting. Her extensive experience provides her with a deep understanding of clinical trial execution from multiple perspectives—site, sponsor, and operational leadership. Deb is known for her clear, direct communication style and exceptional attention to detail, qualities that help her build strong, personalized relationships with clients and cross functional partners. Her leadership at Dartmouth College, where she managed research nurses and study coordinators, has shaped her dedication to operational excellence, team development, and high quality study delivery. Her background includes oversight of both pharmaceutical and medical device programs, with specialized expertise in cardiovascular therapeutic areas. Deb holds a Bachelor of Science degree in Finance and earned her Medical Coding certification from Franklin Pierce University.

Debbie Cabe

Debbie serves as Senior Manager of Clinical Operation at HCC, leading the Operations team composed of CRAs, PCs and Field Clinical Specialists. She comes with over 20 years of clinical research experience across sites, pharmaceutical and medical device industry and CRO environments. She has experience as a CRA and CTM. Debbie brings HCC strong communication skills, and sharp attention clients’ needs and requirements to create a positive personal relationship with a client. Debbie’s experience managing CTM teams at IQVIA Biotech has created a delivery of operational excellence. Debbie has a strong background in both pharmaceutical and medical device projects, majority of experience with Cardiovascular (CAD, vessel closure and coronary & peripheral stenting) Carotid (carotid stenting and embolic protection). Debbie holds a degree in Nursing and Legal Nurse Consultant. Also an active member of ACRP since 2006 , along with a CCRA certification.

Chris Valentine

Chris serves as Senior Manager of Clinical Operations at HCC, leading the Operations team composed of CRAs, PCs and Field Clinical Specialists. He comes with him over 30 years of clinical research experience across the site, pharmaceutical and medical device industry and CRO settings. He has experience as a research coordinator, CRA, PM, in Quality and various operational management roles including an expat assignment in Brussels, Belgium leading an EU clinical operations team. Chris brings to HCC a clear, direct communication style and a strong attention to detail that helps him build meaningful, personalized connections with clients. His leadership experience managing CRA and CTM teams at Abbott Vascular and IQVIA Biotech has shaped his commitment to operational excellence and high quality delivery. Chris’s background spans both pharmaceutical and medical device programs, with deep expertise in Cardiovascular (CAD, stenting, vessel closure) and Carotid (carotid artery stenting and embolic protection) therapeutic areas. Chris holds a BS degree in Biology and Medical Technology from the University of Central Arkansas. He has been an active member of ACRP since 2003 and is dual certified as a CCRA and CCRC.

Joel Meyer

Joel has nearly two decades of experience supporting healthcare physical security IT in operational and clinical environments. He began his career at Salem Health, advancing through multiple leadership roles where he supported hospital-wide operations, managed access control and security systems, supervised teams, and developed workflows, policies, and security training programs. Currently the Manager of Information Technology at Hart Clinical Consultants, Joel oversees IT infrastructure, staff, strategy, security, and vendor relations. He specializes in healthcare-adjacent IT operations, secure infrastructure management, regulatory-aware technology strategy, and aligning technical solutions with organizational needs. Joel holds a Bachelor of Science in Information Technology from Western Governors University.

Ted Prukop

Ted is Director of Business Development at HCC, where he expands partnerships with existing clients and identifies and supports early-stage medical device companies entering the clinical trial pathway. He brings 30+ years of successful leadership experience across sales, sales management, and business development, with a focus on novel, cutting-edge interventional and surgical medical devices. Over his career, Ted has successfully launched several innovative products in the cardiovascular, peripheral, and neurovascular spaces, with roles at Microvena, CHF Solutions, RADI Medical, Angiotech, TriSalus Life Sciences, BTG International, Opsens Medical, and Safeheal. At HCC, he drives long-term value and strategic growth through market research and analysis, lead generation, and strong client relationship management. Ted holds an MBA from Chapman University and a BS degree in Business Administration from Cal Poly, San Luis Obispo.

René Spaargaren, MD


Bruce Gray, DO

Dr. Bruce Gray is a recently retired vascular interventionalist and Professor Emeritus at the University of South Carolina School of Medicine, Greenville. In this role, he provides oversight and mentorship to physician residents and fellows in vascular medicine, ensuring that vascular diagnosis, treatment, and interventional procedures are performed with thoroughness and the highest clinical standards. Dr. Gray also serves as a key member of the HCC Medical Affairs team, offering clinical oversight for peripheral and endovascular initiatives. His expertise informs the development of clinical strategies—shaping subject populations, inclusion and exclusion criteria, and primary and secondary endpoints—to ensure alignment with current clinical practice. His guidance helps ensure that new procedures and emerging peripheral and endovascular technologies are assessed using rigorous scientific methodology balanced with real‑world clinical considerations. Dr. Gray earned his undergraduate degree in Chemistry from The College of Wooster, followed by a master’s degree in Biochemistry from The Ohio State University. He received his medical degree from the Ohio University College of Osteopathic Medicine. He completed his internship and residency in internal medicine at Doctors Hospital in Columbus, Ohio, followed by a vascular medicine fellowship and an interventional vascular medicine fellowship at the Cleveland Clinic.

Chris Metzger, MD

.png)
Rishi Gupta, MD
.png)
Dr. Rishi Gupta is an active, practicing neurointerventionalist who serves as the Co‑Director of the Neuroscience Service Line and Director of Cerebrovascular Care at WellStar Health System in the Atlanta, Georgia region. In these roles, he leads strategic growth initiatives for neuroscience services and works to enhance operational efficiency across departmental programs while meeting key fiscal and performance goals. Dr. Gupta is also a vital member of the HCC Medical Affairs team, where he provides clinical oversight for neurology and neurointerventional projects. His expertise guides the development of clinical strategies, ensuring that subject populations, inclusion and exclusion criteria, and primary and secondary study endpoints are aligned with current clinical practice. His contributions help ensure that innovative procedures and emerging neurovascular devices are evaluated using both rigorous scientific standards and practical, real‑world considerations. Dr. Gupta earned his undergraduate degree from Columbia University’s School of Engineering and Applied Science. He received his medical degree from Meharry Medical College. He completed his neurology residency at Columbia University, followed by fellowships in Neurocritical Care and Stroke Intervention at the Cleveland Clinic, and in Neuroendovascular Intervention at the University of Pittsburgh.
Global Reach
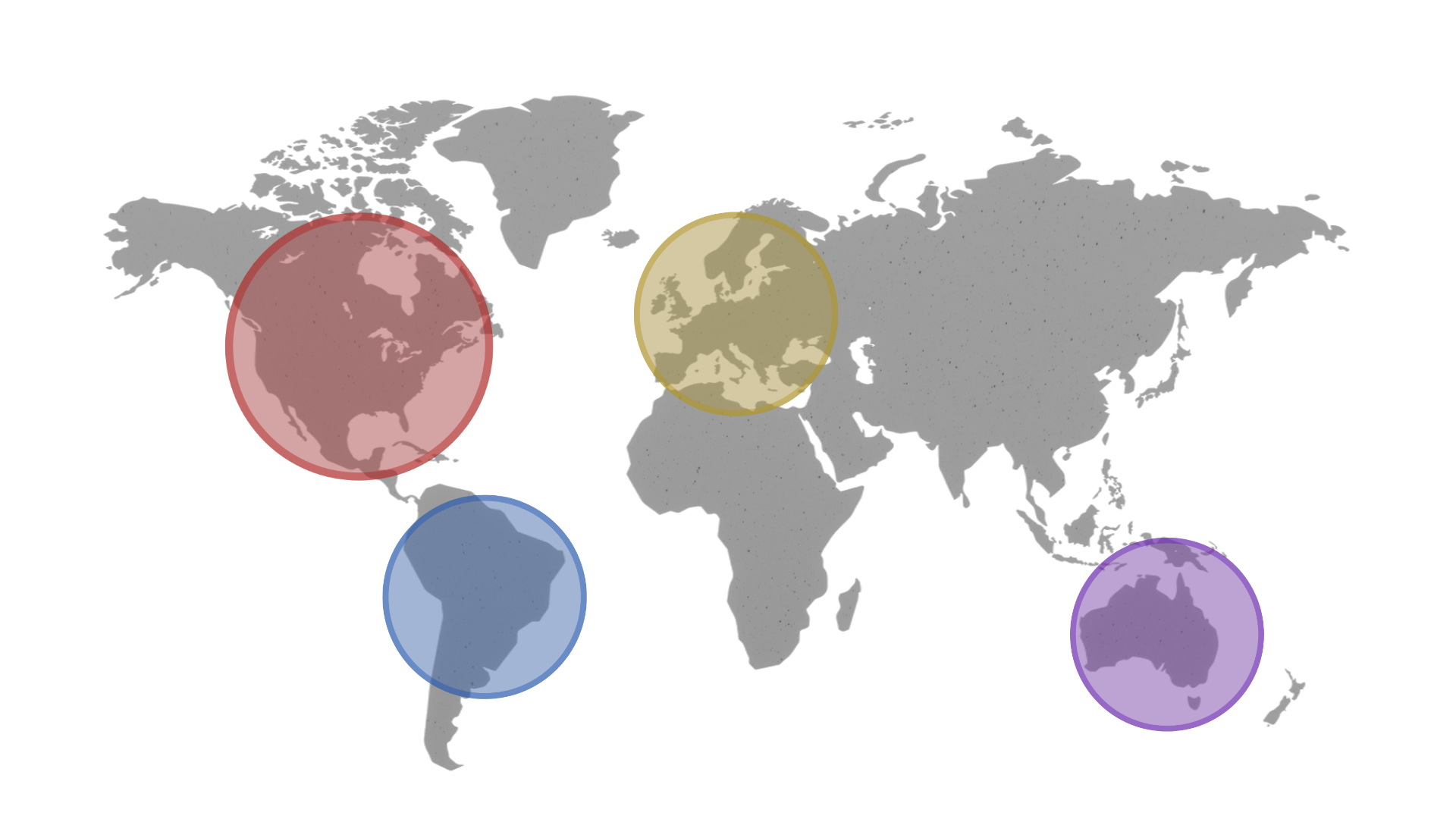
Key moments that shaped HCC
Each milestone marks a step forward in our commitment to rigorous research. These moments define who we are and what we stand for.

