Full Service CRO
Clinical Strategy Development
The success of any project begins with a well‑designed study plan that seamlessly aligns scientific goals with operational needs. HCC’s broad network of physicians and interventional specialists collaborates closely with entrepreneurs and technology developers to ensure real‑world scenarios are prioritized throughout the study lifecycle. This proactive approach enables teams to anticipate potential challenges and implement clear, effective strategies that enhance operational efficiency while helping to control both budget and timeline demands.
- Clearly defined study objectives, supported by a focused, purpose‑built strategy
- An adaptive, execution‑ready protocol approach designed to evolve as the study progresses
- Patient‑centric enrollment strategies that drive efficiency and improve study performance
- Continuous assessment of risks and progress, with timely adjustments as required
.png)

Regulatory Affairs/
FDA Submission Support
HCC develops a comprehensive strategy, maintains thorough documentation, and engages proactively with regulatory agencies to uphold compliance and facilitate the smooth advancement of your program.
- Regulatory Affairs (Global): Prepare and submit regulatory documents and IRB applications; coordinate with health authorities to enable timely study startup
- Agency Communications: Oversee all communications related to regulatory submissions and pre‑submission meetings; manage updates, safety reporting, and responses to health authority queries
- FDA Submission Support: Develop IND/IDE applications, briefing packages, and pre‑submission materials; manage ongoing FDA interactions, including protocol amendments throughout the trial
- Closeout support: Deliver final regulatory reports, manage post‑submission follow‑ups, and prepare submission‑ready documentation such as final study reports and briefing packages
Project Management
HCC’s Project Managers are equipped with extensive therapeutic and research expertise, enabling them to foresee and address potential project challenges effectively. This ensures that your project is completed within the stipulated timeframe and budget constraints.
- Study Startup Leadership: Oversee feasibility, regulatory submissions, and contract management to ensure sites are activation‑ready
- Trial Execution Oversight: Guide site performance, enrollment activities, monitoring operations, and vendor coordination throughout the study
- Closeout Coordination: Manage final data reconciliation, site closures, and preparation of deliverables that support regulatory submissions
- Proactive Communication: Foster alignment across teams and maintain clear, timely communication to help deliver the study on schedule and within budget
.png)

Site Management
Our team uses advanced platforms that follow agency filing and naming standards to manage and store clinical trial documents efficiently. The team ensures accurate and timely payments to clinical sites by maintaining proactive communication with site and sponsor staff. HCC also manages investigational inventory, tracking the chain of custody for devices or agents throughout their lifecycle. Leveraging extensive clinical site data, we provide recommendations and referrals that support rapid study startup, strong enrollment, and high-quality data.
- Strategic Site Selection & Activation: Identify and activate the most suitable sites through targeted feasibility assessments and close collaboration with site staff
- Hands‑On Site Support: Provide ongoing support for visit scheduling, enrollment tracking, and oversight of protocol compliance
- Rapid Issue Resolution: Maintain frequent, proactive communication to resolve issues quickly and keep sites engaged, informed, and compliant.
- Clean, Audit‑Ready Closeout: Ensure complete and compliant documentation, along with robust support for final data collection and audit readiness.
Study Monitoring
HCC assigns study monitors with intention and precision, selecting individuals whose therapeutic expertise aligns with each project’s unique requirements. This targeted approach ensures that study data is evaluated with a discerning eye, strengthening protocol adherence, enhancing data accuracy, and supporting thorough safety event reporting. HCC monitors also serve as dependable extensions of the study team, providing comprehensive training and remediation whenever needed.
In addition, HCC incorporates geographic alignment into monitor assignments to promote efficient site coverage, reduce travel‑related delays, and reinforce existing site relationships-further enhancing the consistency and quality of site support.
In addition, HCC incorporates geographic alignment into monitor assignments to promote efficient site coverage, reduce travel‑related delays, and reinforce existing site relationships-further enhancing the consistency and quality of site support.
- Early Monitor Engagement: Engage monitors from the outset to support site training, participate in investigator meetings, and contribute to the development of tailored monitoring plans
- Comprehensive Monitoring Approach: Conduct consistent on‑site and remote monitoring visits to verify source data, assess protocol compliance, and ensure the well‑being of enrolled study participants
- Risk‑Based, Flexible Focus: Prioritize critical data and processes while maintaining the flexibility to adapt in real time to site‑specific or study‑specific needs
- Thorough Closeout Support: Deliver final data verification, timely query resolution, and audit‑ready documentation to ensure a clean study closeout and readiness for regulatory inspections


Clinical Safety Management
HCC’s Safety Management team provides comprehensive clinical safety oversight throughout the clinical study lifecycle. Our experts handle adverse event review, safety reporting, and signal evaluation in line with FDA and ICH GCP standards. We coordinate Clinical Events Committees and Data Safety Monitoring Boards for rigorous oversight and reliable safety data. Led by our medical director, HCC ensures strict adherence to the Safety Management Plan for compliance and operational excellence tailored to your project needs.
- Safety Management Plans & Readiness: Establish clear reporting procedures and fully prepared systems to support efficient and accurate adverse event tracking
- Adverse Event Reviews: Conduct ongoing evaluations of seriousness and causality, ensuring consistent, compliant, and well‑documented assessments throughout the study
- CEC / DSMB Management: Oversee charter development, meeting coordination, documentation, and final committee reporting and archiving to ensure structured, compliant safety oversight
- Safety Reporting: Provide comprehensive support for expedited safety reporting, reconciliation activities, and documentation to facilitate regulatory submissions and inspection readiness
Biometrics
HCC provides disciplined processes and seasoned oversight to ensure your datasets are clean, analysis‑ready, and properly prepared for regulatory review. Key capabilities include:
- Data Management: Develop CRFs, build and validate edit checks, conduct user acceptance testing, manage queries, generate metrics and reports, produce data exports, oversee database lock, and ensure compliant data archival
- Medical Coding & Reconciliation: Perform AE/SAE reconciliation and apply standardized coding using MedDRA and WHODrug (as applicable)
- Biostatistics: Develop statistical sections of the protocol, support randomization and sample‑size calculations, create SAPs, execute analyses and reporting, and contribute to regulatory submission packages
- Submission‑Ready Deliverables: Produce high‑quality TLFs and CDISC‑compliant SDTM/ADaM datasets accompanied by complete, submission‑ready documentation
.png)

Site & Sponsor BIMO Inspection Preparedness
HCC’s certified clinical auditors partner with sponsors and clinical sites to ensure comprehensive preparedness for agency inspections, helping to minimize the impact of inspection findings on investigational product approval.
- Mock BIMO Inspection Audits: Conduct simulated inspection exercises to evaluate inspection‑hosting capabilities, test operational readiness, and strengthen team preparedness
- Standard BIMO Preparation Audits: Perform comprehensive records‑focused audits to identify gaps, ensure documentation accuracy, and maintain continuous inspection readiness
- Targeted / Directed Audits: Execute focused audits—upon sponsor request—to verify specific delegated activities and ensure they meet regulatory and study requirements
- Core Focus Areas: Assess adherence to subject rights, safety, and welfare; confirm data accuracy and reliability; and evaluate protocol and regulatory compliance across all audited processes
Technical Writing
HCC provides expert medical writing support across the clinical development lifecycle. Our team prepares protocols, investigator brochures, clinical study reports, and safety narratives aligned with FDA regulations and ICH guidelines. Combining scientific expertise with regulatory insight, HCC ensures complex clinical data are clearly communicated and positioned to support regulatory submissions.
- Interim Reports & Clinical Study Reports (CSRs): Develop clear, comprehensive interim reports and full CSRs to support key study milestones and regulatory submission packages
- Annual Reports: Prepare annual reports for the FDA and other regulatory authorities, as required by sponsor obligations and regional regulations
- Submission‑Ready Documentation: Produce documentation tailored for regulatory use cases-including annual updates and study‑specific submissions-to ensure completeness, accuracy, and compliance

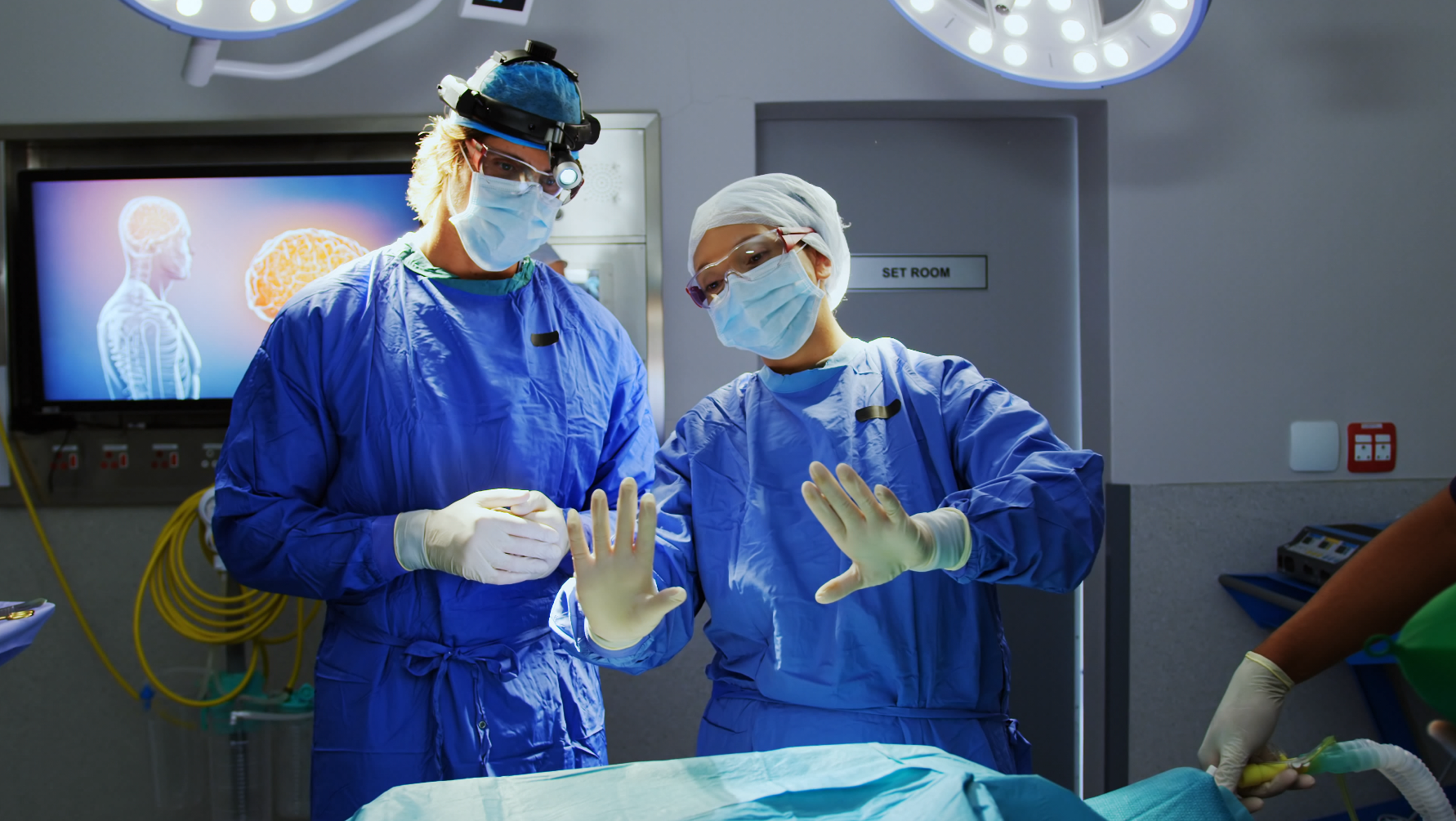
Field Clinical Specialist
HCC’s Field Clinical Specialists (FCS) deliver high‑touch clinical support to ensure innovative procedures are performed in full compliance with instructions for use (IFU) and study protocols, and that novel medical devices are properly prepared and utilized. Working seamlessly in both cath lab and OR environments, FCS's provide hands‑on oversight that helps prevent procedural errors and ensures accurate collection of essential procedural data.
- Case Proctoring: Develop comprehensive case proctoring plans, procedural standards, and readiness activities to ensure consistent, compliant execution across sites
- Product Training: Deliver hands-on training to site teams on investigational product administration, handling, storage, and accountability
- Enrollment Enhancement: Collaborate with investigators and coordinators to enhance patient screening and ensure data accuracy and compliance with protocols
